Basic Types of Radiation: Alpha, Beta & Gamma


Ignorance of the radiation causes fear, and there is little knowledge in the prepping world as a whole about the different types of radiation.
Few things cause as much fear as radiation. Sci-Fi movies use radiation as a plot device. Radiation was the cause for Godzilla, the Hulk, the 50 foot woman, Spider man, even Superman.
Radiation is like congress and spending cuts, it sounds good on TV but the facts are much different than what you see.
In fact, as a hazardous material technician, I would MUCH rather deal with a radiological release than any other hazardous material for several reasons.
I’d Rather Deal with Radiation the Hazardous Materials
- Radiological contamination is much easier to find with instruments than the typical hazardous chemical.
- Due to the nature of radiological materials, I can protect myself with a basic knowledge of chemistry. The inverse square law shops how close is too close.
- We can also create shielding to protect against radiological exposure. A dust mask and paper coveralls is sufficient to protect against most common radiological spills.
- Rain and other environmental factors can make a chemical spill much worse. For example moisture can turn chlorine gas into hydrochloric acid or hypochlorous acid; where as a radiological material’s properties are not changed by the weather making it a lot more predictable.
However, the purpose of this article is not to be a cheerleader for radiation, but rather to give a simple explanation of the types of radiation most commonly encountered by a citizen.
What is Radiation?
Basically radiation is energy traveling through material. Light, heat, radio and microwaves are all radiating out from their sources. Radiation is all around you, but that radiation is divided into two basic classes, ionizing and non-iodizing radiation.
If the radiation that has enough energy to knock around atoms in a molecule and cause them to vibrate, but not enough to actually kick electrons out of the molecule, is referred to as “non-ionizing radiation”. Non-ionizing radiation examples include visible light and microwaves.
If the radiation is powerful to bounce electrons out from their atoms it is “ionizing” because it creates ions. When we thing our “radiation” this is the type we normally thing about.
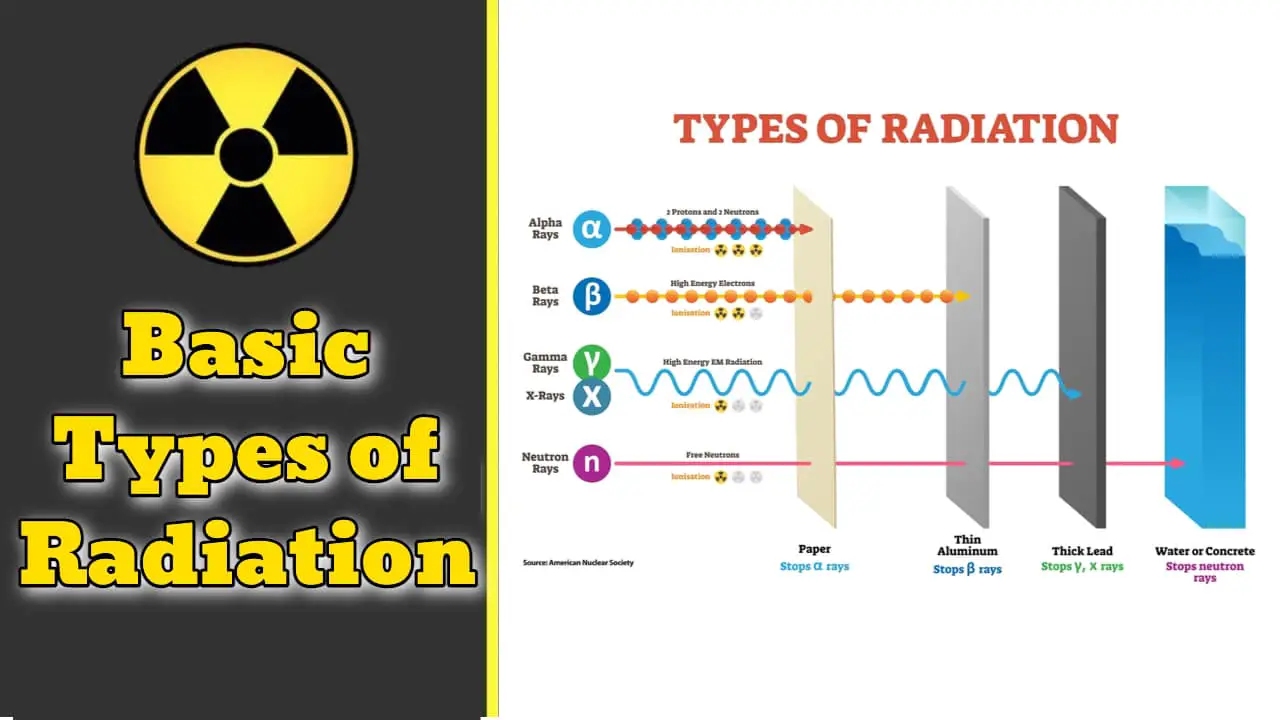
There are several different types of radiation, but the three most related to our health and the most common are Alpha, Beta and Gamma radiation.
Alpha Radiation
Alpha radiation is a heavy, very short-range particle and is actually an ejected helium nucleus. Some characteristics of alpha radiation are:
- Alpha radiation is not able to penetrate human skin.
- Alpha-emitting materials can be harmful to humans if the materials are inhaled, swallowed, or absorbed through open wounds.
- A thin-window Geiger-Mueller (GM) probe can detect the presence of alpha radiation. (This is normally called a pancake probe as it is flat and round)
- Most other Instruments cannot detect alpha radiation because even a thin layer of water, dust, paper, or other material, because alpha radiation is not penetrating.
- Alpha radiation travels only a short distance (a few inches) in air, but is not an external hazard.
- Alpha radiation is not able to penetrate clothing
Examples of some alpha emitters: radium, radon, uranium, thorium.
Beta Radiation
Beta radiation is a light, short-range particle and is actually an ejected electron. Some characteristics of beta radiation are:
- Beta radiation may travel several feet in air and is moderately penetrating.
- Beta radiation can penetrate human skin to the “germinal layer”. New skin cells grow at the germinal layer. An injury known as a “Beta Burn” occurs if beta-emitting contaminants are on the skin for a prolonged period of time
- Beta-emitting contaminants may be harmful if deposited internally.
- Survey instruments with a pancake probe are detect beta. Some beta emitters, however, produce very low-energy, poorly penetrating radiation that may be difficult or impossible to detect. Examples of these difficult-to-detect beta emitters are hydrogen-3 (tritium), carbon-14, and sulfur-35.
- Clothing provides protection against beta radiation
Examples of some pure beta emitters: strontium-90, carbon-14, tritium, and sulfur-35.
Gamma Radiation
Gamma radiation is highly penetrating electromagnetic radiation. Some characteristics of this radiations is:
- Gamma radiation are able to travel many feet in air and many inches in human tissue. Gamma will penetrate most materials. This types is also known as “penetrating” radiation.
- Gamma radiation are electromagnetic radiation like visible light, radio-waves, and ultraviolet light. These electromagnetic radiations differ only in the amount of energy they have. Gamma rays are the most energetic of these.
- It requires dense shielding materials. Clothing provides little shielding from penetrating radiation, but will prevent contamination of the skin by gamma-emitting radioactive materials.
- Survey meters with a sodium iodide detector probe easily detect Gamma radiation. (Called a “hotdog” because it is long and cylindrical”
- Gamma radiation frequently accompany the emission of alpha and beta radiation during radioactive decay.
Examples of some gamma emitters: iodine-131, cesium-137, cobalt-60, radium-226, and technetium-99m.